A*STAR NEWS
Singapore Scientists Dissect Rna Structures Of Sars-cov-2 And Discover Important Host Factor For Infection
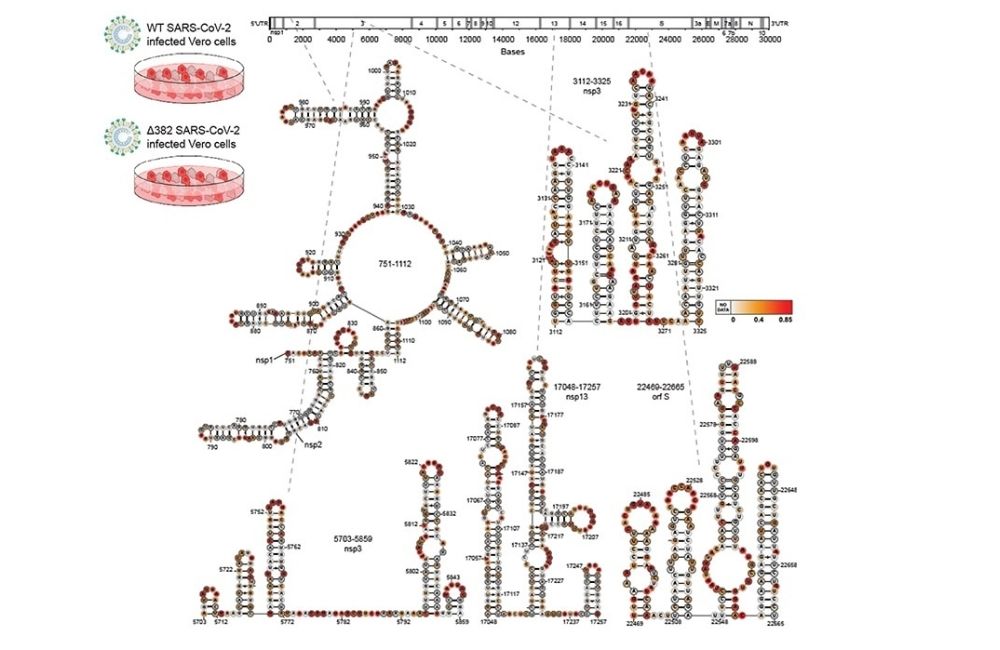 SARS-CoV-2 genome folds into complex structures inside host cells.
SARS-CoV-2 genome folds into complex structures inside host cells.
A team of researchers from Agency for Science, Technology and Research (A*STAR)’s Genome Institute of Singapore (GIS) and Bioinformatics Institute (BII), together with Duke-NUS Medical School, integrated a variety of technologies to study not only the whole genome of SARS-CoV-2, but also its RNA structures, and discovered an important host factor for infection. Additionally, they also mapped the shapes of a deletion variant that was isolated from a patient in Singapore, allowing them to determine the structures of both wildtype and other variants. The study was published in Nature Communications on 25 August 2021 (DOI: 10.1038/s41467-021-25357-1).
SARS-CoV-2, which causes COVID-19, is an RNA virus whereby its genome is made up of a single 30 kilobases long RNA. This long RNA can fold into complex and dynamic shapes to allow the virus to interact with other factors for virus growth and survival inside infected cells. While it is traditionally difficult to determine RNA shapes of long viruses, GIS scientists used a chemical to mark the positions of all single-strand bases along the genome, and then identified them using sequencing. The scientists identified which regions are paired / unpaired, and integrated this information into computational models to determine more accurate RNA shapes.
Through this study, they discovered that the virus’ genome can fold into very complex shapes and interact with many other of its host’s RNAs. One such RNA is a small RNA known as snoRNA, which normally modifies our translation machinery to enable our body to produce protein properly. The team discovered that the SARS-CoV-2 also binds with snoRNA to steal its modification. This enables it to decrease the amount of host RNAs, as well as become more stable and successful in infecting its host’s cells.
Dr Wan Yue, Group Leader of Laboratory of RNA Genomics and Structure, and Associate Director of Epigenetic and Epitranscriptomic Systems at GIS, said, “When SARS-CoV-2 emerged and COVID-19 became pandemic, we raced to understand how SARS-CoV-2 can fold into shapes and interact with host RNAs for their infectivity. We found that SARS-CoV-2 genome interacts with snoRNAs to compete with host RNAs for methylation, to stabilise its own genome and to degrade the host RNA. Furthermore, people have increasingly realised in recent years that RNA shapes, similar to protein shapes, can be targeted by small molecules and our mapping of SARS-CoV-2 RNA shapes could be useful for future drug development.”
Prof Patrick Tan, Executive Director of GIS, said, “SARS-CoV-2 is the major pathogen all over the world now. Understanding how it infects human cells is key to our ability to inhibit it. We believe that the shape maps that were generated in this study can serve as a basis for drug targeting of this virus in the future.”
This study was carried out together with Dr Roland Huber (Assistant Principal Investigator at A*STAR’s BII) and Prof Wang Linfa (Professor, Emerging Infectious Diseases Programme, Duke-NUS Medical School).
- END -About A*STAR’s Genome Institute of Singapore (GIS)
The Genome Institute of Singapore (GIS) is an institute of the Agency for Science, Technology and Research (A*STAR). It has a global vision that seeks to use genomic sciences to achieve extraordinary improvements in human health and public prosperity. Established in 2000 as a centre for genomic discovery, the GIS pursues the integration of technology, genetics and biology towards academic, economic and societal impact, with a mission to "read, reveal and write DNA for a better Singapore and world".
Key research areas at the GIS include Precision Medicine & Population Genomics, Genome Informatics, Spatial & Single Cell Systems, Epigenetic & Epitranscriptomic Regulation, Genome Architecture & Design, and Sequencing Platforms. The genomics infrastructure at the GIS is also utilised to train new scientific talent, to function as a bridge for academic and industrial research, and to explore scientific questions of high impact.
For more information about GIS, please visit www.a-star.edu.sg/gis.
About the Agency for Science, Technology and Research (A*STAR)
A*STAR is Singapore's lead public sector R&D agency. Through open innovation, we collaborate with our partners in both the public and private sectors to benefit the economy and society. As a Science and Technology Organisation, A*STAR bridges the gap between academia and industry. Our research creates economic growth and jobs for Singapore, and enhances lives by improving societal outcomes in healthcare, urban living, and sustainability. A*STAR plays a key role in nurturing scientific talent and leaders for the wider research community and industry. A*STAR’s R&D activities span biomedical sciences to physical sciences and engineering, with research entities primarily located in Biopolis and Fusionopolis. For ongoing news, visit www.a-star.edu.sg.
ANNEX B
The research findings described in this media release can be found in the scientific journal Nature Communications, under the title, “Comprehensive mapping of SARS-CoV-2 interactions in vivo reveals functional virus-host interactions” (DOI: 10.1038/s41467-021-25357-1) by the following authors: Siwy Ling Yang1, Louis DeFalco2, Danielle E. Anderson3, Yu Zhang1, Ashley J Aw1, Su Ying Lim1, Xin Ni Lim1, Kiat Yee Tan1 Tong Zhang1, Tanu Chawla3, Yan Su4, Alexander Lezhava4, Andres Merits5, Lin-Fa Wang3*, Roland G. Huber2*, Yue Wan1,6,7*
1. Epigenetic and Epitranscriptomic Regulation, Genome Institute of Singapore, Agency for Science, Technology and Research (A*STAR), Singapore 138672, Singapore
2. Biomolecular Function Discovery, Bioinformatics Institute (BII), Agency for Science, Technology and Research (A*STAR), Matrix #07-01, Singapore 138671
3. Programme in Emerging Infectious Diseases, Duke-NUS Medical School, 8 College Road, 169857, Singapore
4. Laboratory of translational diagnostics, Genome Institute of Singapore, Agency for Science, Technology and Research (A*STAR), Singapore 138672, Singapore
5. Institute of Technology, University of Tartu, Tartu, Estonia
6. School of Biological Sciences, Nanyang Technological University, Singapore 637551
7. Department of Biochemistry, Yong Loo Lin School of Medicine, National University of Singapore, Singapore 117597, Singapore.
These authors contributed equally: Siwy Ling Yang, Louis DeFalco, Danielle E. Anderson, Yu Zhang, Ashley J Aw.
These authors jointly supervised this work: Lin-Fa Wang (linfa.wang@duke-nus.edu.sg ), Roland Huber (rghuber@bii.a-star.edu.sg ), Yue Wan (wany@gis.a-star.edu.sg ).
Was This Article Helpful ?
A*STAR celebrates International Women's Day

From groundbreaking discoveries to cutting-edge research, our researchers are empowering the next generation of female science, technology, engineering and mathematics (STEM) leaders.
Get inspired by our #WomeninSTEM
