Singapore scientists and physicians awarded prestigious NRF grant to develop novel therapies for liver cancer
The team aims to develop preventive interventions for liver cancer using innovative, state-of-the-art genomic technology.
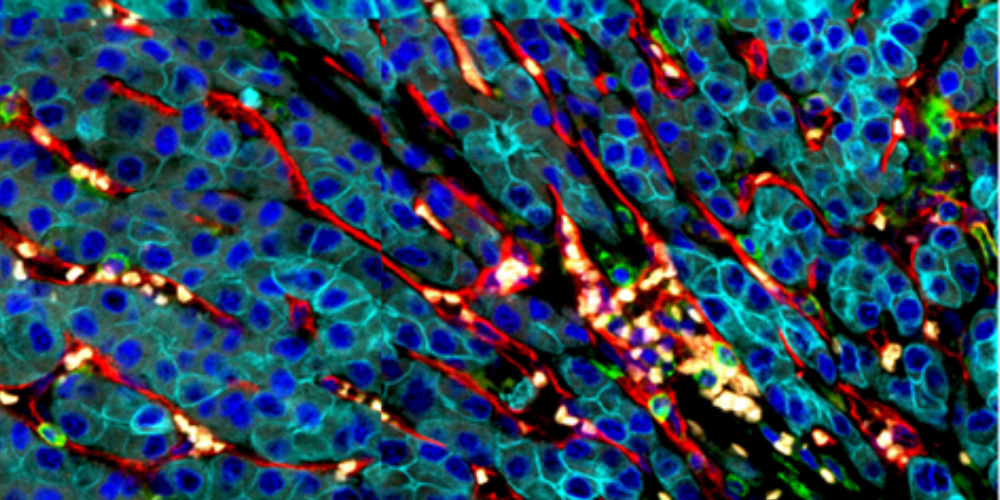 Microscopic image of a tumour (cancer cells stained in cyan) being infiltrated with immune cells (green) that are recruited through blood vessels (red). Individual cell nuclei stained in blue. (Image credit: A*STAR’s GIS)
Microscopic image of a tumour (cancer cells stained in cyan) being infiltrated with immune cells (green) that are recruited through blood vessels (red). Individual cell nuclei stained in blue. (Image credit: A*STAR’s GIS)
SINGAPORE – A team of multidisciplinary scientists has been awarded the prestigious National Research Foundation (NRF) Singapore grant to uncover the fundamental mechanisms of liver disease progression from fatty liver disease to fibrosis/cirrhosis (liver scarring), and eventually, liver cancer.
The team aims to identify novel therapeutic targets and biomarkers that can be developed into new therapies to prevent progression to cancer, as well as biomarkers that can be used to detect and monitor early disease progression to allow prompt preventive interventions.
The research is led by A*STAR’s Genome Institute of Singapore (GIS), in collaboration with A*STAR’s Singapore Immunology Network (SIgN), Yong Loo Lin School of Medicine, National University of Singapore (NUS Medicine), National University Hospital (NUH) and Cancer Science Institute (CSI), SingHealth Duke-NUS Translational Immunology Institute (TII), and Nanyang Technological University Singapore’s (NTU Singapore) School of Biological Sciences.
Hepatocellular carcinoma (HCC), the most common type of primary liver cancer in adults, is also one of the most prevalent cancers in the Asia-Pacific region, including Singapore. In terms of cancer deaths in Singapore, it was ranked third among men and fourth among women from 2011-20151 . HCC arises in chronically damaged, and inflamed livers with the underlying disease progression caused by viral hepatitis, and non-viral factors such as alcohol-mediated damage and fatty liver disease. Prolonged inflammation associated with chronic non-healing wounds can lead to the formation of cancer.
Additionally, a recent study in Cell by the GIS Laboratory of Precision Oncology and Cancer Evolution revealed that early developmental programmes can be reactivated in both the tumour as well as other inflammatory disease-associated microenvironment, such as cirrhosis. These observations suggest that there are “dormant embryonic programmes” that can be harnessed to promote wound healing and tissue repair. The appreciation for the role of the tissue environment in chronic, non-healing wounds, is essential for the understanding of the biology and evolution of tumour growth.
The team is now motivated to understand the early mechanisms that underlie the progression of early liver disease to advanced fibrosis or cirrhosis, and eventually to HCC. The primary research objective of the project is to generate a comprehensive single-cell catalogue of the evolution of disease-associated cell states (both in liver epithelium and tissue environment) that control the trajectories of the disease progression to HCC.
Dr Ramanuj DasGupta, Senior Group Leader of the Laboratory of Precision Oncology and Cancer Evolution at GIS, and Lead PI of the HCC project said, “Our research is focused on understanding how tumours evolve and hijack programmes of gene expression that are similar to those used during fetal development and wound healing in order to escape detection by the body’s own immune system. Our goal is to reactivate the body’s immune surveillance to identify these tumour cells as ‘foreign’, and eliminate them.”
Associate Prof Dan Yock Young, Department of Medicine, NUS Medicine and Senior Consultant, Division of Gastroenterology & Hepatolgy, NUH said, “Prevalence of non-alcoholic fatty liver disease is growing in epidemic proportions and is poised to become the commonest cause of liver cancer worldwide. While much effort has focused on how to better treat cancer when it develops, this grant allows us to move upstream to understand why and how liver cancer develops along the injury process with the intent of preventing it before it develops.”
Dr Valerie Chew from SingHealth Duke-NUS TII, a senior HCC immunologist, and team member of the project, said, “As the immune milieu of HCC is very complex and plays an important role in response to immunotherapy, I believe that a better understanding of cross-talk among various immune populations as well as their interactions with the tumour cells within the microenvironment will lead to a breakthrough in the treatment of HCC.”
Prof Patrick Tan, Executive Director of GIS, said, “Discoveries from the study will aid the development of new therapeutics and treatment paradigms designed at targeting HCC. We believe that this collaborative network of domain experts, combined with unique cross-sectional clinical samples, will help bring our research objectives to fruition, and eventually make a difference in the clinic to benefit patients.”
– END –1 https://www.nuh.com.sg/Health-Information/Diseases-Conditions/Pages/Hepatocellular-Carcinoma.aspx
A*STAR celebrates International Women's Day

From groundbreaking discoveries to cutting-edge research, our researchers are empowering the next generation of female science, technology, engineering and mathematics (STEM) leaders.
Get inspired by our #WomeninSTEM
