A*STAR NEWS
High-Resolution Genome Mapping Lays Groundwork in Fight Against Antibiotic Resistant Gut Bacteria
SINGAPORE – A study led by scientists from the Agency for Science- Technology and Research’s (A*STAR) Genome Institute of Singapore (GIS)- together with clinicians from Tan Tock Seng Hospital (TTSH) and researchers from the National University of Singapore (NUS)- has resulted in a breakthrough approach to studying gut bacteria- and a step forward in the battle against antibiotic resistance.
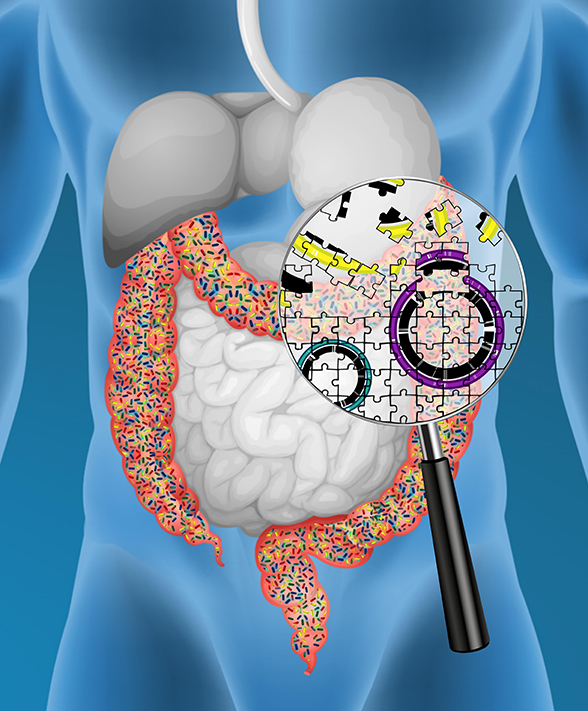
Genome reconstruction of the gut bacteria as a complex genetic puzzle
(Copyright: A*STAR’s Genome Institute of Singapore)
Their technique leverages portable DNA sequencing technology and sophisticated clustering algorithms to analyse complex bacterial communities in the gut- pulling together their entire genetic code to track the spread of antibiotic resistance and study microbial contributions to human health. The paper was published in Nature Biotechnology on 29 July 2019.
Antibiotic-resistant microorganisms have become a major threat to healthcare systems worldwide- estimated to impact more than 10 million lives annually and cost the global economy more than 100 trillion US dollars by 2050[1].
The human gut harbours trillions of bacteria that benefit the host in various ways - from breaking down undigested food- to producing essential vitamins and nutrients- and training the immune system. However- gut bacteria can also serve as a ‘reservoir’ for antibiotic-resistant bacteria- allowing for long-term asymptomatic colonisation and transfer of resistance to other bacteria.
Until now- studies have been limited by the resolution of genetic analysis and the ability to understand microbial function in communities harbouring hundreds of bacterial species. The newly-developed method provides a more complete genomic map allowing scientists to study how individual bacterial strains contribute to overall microbial community function in the human body.
In order to develop this method- the research team led by Prof Niranjan Nagarajan and Dr Denis Bertrand from GIS experimented with a range of technologies. They finally narrowed down their search to a portable DNA nanopore sequencer that provides long DNA 'reads'[2] and high data throughput.
By assessing 197 stool samples from ongoing clinical studies- they refined and established a protocol that could analyse a majority of the samples and provide high-quality data. By combining this data with accurate short reads- the researchers developed a hybrid assembly algorithm- OPERA-MS- that pieces together billions of DNA sequences into individual strain genomes present in the bacterial community. OPERA-MS combines a sophisticated Bayesian clustering algorithm with combinatorial optimisation to solve the extremely challenging DNA puzzles that ‘metagenomes’[3] represent.
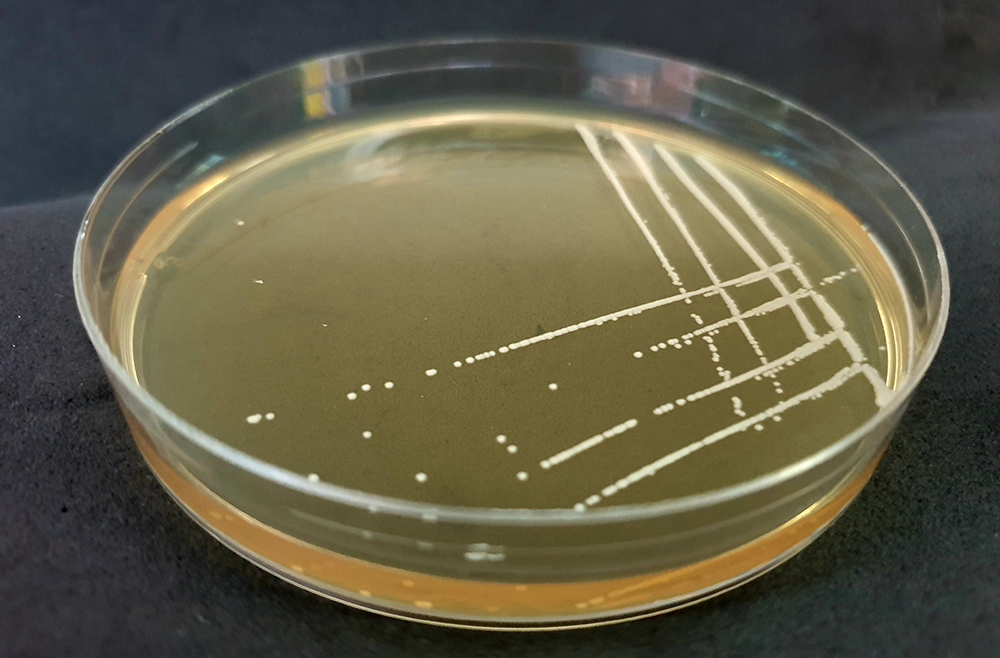
Cultures of antibiotic resistant Klebsiella pneumoniae from stool samples
(Copyright: A*STAR’s Genome Institute of Singapore)
Benchmarking this new approach against existing methods- researchers found that OPERA-MS provides up to 10 times more complete genomes compared to short reads- and at least 5 times more accurate genomes than when using long reads.
Prof Pavel Pevzner- Ronald R. Taylor Chair and Distinguished Professor of Computer Science at University of California San Diego- said- “Although shotgun metagenomics revolutionized analysis of microbial communities- short- read metagenomic assemblies are typically very fragmented. Nagarajan and colleagues demonstrated that the recently emerged long-read technologies- that have already opened a new era in analyzing isolate genomes- have now opened a new era of “contiguous assemblies” in metagenomics. I now expect an explosion of long-read metagenomic studies since the contiguity of assemblies is a pre-requisite for many follow-up studies such as antibiotics discovery from metagenomes.”
Dr Denis Bertrand- Staff Scientist at GIS and lead author of this work said- “Hybrid metagenomic assembly with OPERA-MS provides unprecedented resolution at the strain level and near-complete genomes from uncultured samples. We believe this approach can serve as the basis of gold-standard metagenome-assembled genomes for future studies.”
Dr Adam Phillippy- Investigator at the U.S. National Human Genome Research Institute- said- “Recovering high-quality microbial genomes from metagenomic samples is a notoriously hard problem that demands clever solutions. OPERA- MS combines multiple clever solutions- leveraging both short and long-read sequencing- to assemble accurate and continuous genomes out of metagenomes. Such assemblies will be crucial for understanding the function of complex metagenomes.”
Together with clinicians at TTSH- GIS scientists have been applying this new approach to study the colonisation of the gut by multi-antibiotic resistant bacteria. While the presence of thousands of bacterial species and strains in the human gut may make distinguishing ‘friend from foe’ challenging- GIS and TTSH researchers were able to use OPERA-MS to tease apart genomes of commensal gut strains from newly colonising antibiotic resistant strains to characterise their source and function better.
This allowed them to assemble many completely novel resistance gene combinations. The researchers noted that one of the sequences assembled was the closed genome of a 263-000 base long “jumbo” phage[4] that had never been seen before. This discovery could potentially result in new treatment options for antibiotic-resistant bacteria.
Prof Niranjan Nagarajan- Associate Director and Senior Group Leader at GIS- noted- “The complete genome sequence of an organism is its ultimate fingerprint. We are excited to now be able to extract it directly out of metagenomic data. We need powerful technological capabilities like this to successfully apply clinical metagenomics in the battle against antibiotic resistance.”
Prof Patrick Tan- Executive Director at GIS- said- “It is exciting to see the power of new nanopore sequencing technologies being brought to bear on this important problem. Real-time clinical metagenomics can transform our understanding of how antibiotic-resistant organisms spread and help us curb this growing menace.”
[1] Source: Review on Antimicrobial Resistance
[2] Information on the genetic sequence corresponding to a DNA fragment
[3] Genetic material present in a microbial community e.g. in the human gut
[4] Viruses that infect bacteria and archaea
Notes to Editor:
The research findings described in this media release can be found in the scientific journal Nature Biotechnology- under the title- “Hybrid metagenomic assembly enables high-resolution analysis of resistance determinants and mobile elements in human microbiomes” by Denis Bertrand1- Jim Shaw1- Manesh Kalathiappan1- Amanda Hui Qi Ng1- Senthil Muthiah1- Chenhao Li1- Mirta Dvornicic1-2- Janja Paliska Soldo1- Jia Yu Koh1- Chengxuan Tong1- Ng Oon Tek3- Timothy Barkham4- Barnaby Young3-5- Kalisvar Marimuthu6-7- Chng Kern Rei1- Mile Sikic2- Niranjan Nagarajan1.
1. Computational & Systems Biology- Genome Institute of Singapore- 60 Biopolis Street- #02-01 Genome- Singapore 138672- Singapore
2. Faculty of Electrical Engineering and Computing- Department of Electronic Systems and Information Processing- University of Zagreb- Unska 3- 10000 Zagreb- Croatia
3. National Centre for Infectious Disease- Tan Tock Seng Hospital- 11 Jalan Tan Tock Seng Singapore 308433- Singapore
4. Department of Laboratory Medicine- Tan Tock Seng Hospital- Singapore 308433- Singapore
5. Lee Kong Chian School of Medicine- Nanyang Technological University- 50 Nanyang Ave- Singapore 308232- Singapore
6. Institute of Infectious Diseases and Epidemiology- Tan Tock Seng Hospital- 11 Jalan Tan Tock Seng- Singapore- 304833- Singapore
7. National University of Singapore- 21 Lower Kent Ridge Road- Singapore- 119077- Singapore
* Corresponding Author: Niranjan Nagarajan (nagarajann@gis.a-star.edu.sg)
About A*STAR’s Genome Institute of Singapore (GIS)
The Genome Institute of Singapore (GIS) is an institute of the Agency for Science- Technology and Research (A*STAR). It has a global vision that seeks to use genomic sciences to achieve extraordinary improvements in human health and public prosperity. Established in 2000 as a centre for genomic discovery- the GIS will pursue the integration of technology- genetics and biology towards academic- economic and societal impact.
The key research areas at the GIS include Human Genetics- Infectious Diseases- Cancer Therapeutics and Stratified Oncology- Stem Cell and Regenerative Biology- Cancer Stem Cell Biology- Computational and Systems Biology- and Translational Research.
The genomics infrastructure at the GIS is utilised to train new scientific talent- to function as a bridge for academic and industrial research- and to explore scientific questions of high impact.
For more information about GIS- please visit www.a-star.edu.sg/gis.
About the Agency for Science- Technology and Research (A*STAR)
The Agency for Science- Technology and Research (A*STAR) is Singapore's lead public sector agency that spearheads economic oriented research to advance scientific discovery and develop innovative technology. Through open innovation- we collaborate with our partners in both the public and private sectors to benefit society.
As a Science and Technology Organisation- A*STAR bridges the gap between academia and industry. Our research creates economic growth and jobs for Singapore- and enhances lives by contributing to societal benefits such as improving outcomes in healthcare- urban living- and sustainability.
We play a key role in nurturing and developing a diversity of talent and leaders in our Agency and research entities- the wider research community and industry. A*STAR’s R&D activities span biomedical sciences and physical sciences and engineering- with research entities primarily located in Biopolis and Fusionopolis.
Was This Article Helpful ?
A*STAR celebrates International Women's Day

From groundbreaking discoveries to cutting-edge research, our researchers are empowering the next generation of female science, technology, engineering and mathematics (STEM) leaders.
Get inspired by our #WomeninSTEM