A*STAR NEWS
PRESS RELEASES
Human Heart Tissue Grown from Stem Cells Improves Drug Testing
Singapore – Researchers at the Institute of Bioengineering and Nanotechnology (IBN) of A*STAR have engineered a three-dimensional heart tissue from human stem cells to test the safety and efficacy of new drugs on the heart.
“Cardiotoxicity- which can lead to heart failure and even death- is a major cause of drug withdrawal from the market. Antibiotics- anticancer and antidiabetic medications can have unanticipated side effects for the heart. So it is important to test as early as possible whether a newly developed drug is safe for human use. However- cardiotoxicity is difficult to predict in the early stages of drug development-” said Professor Jackie Y. Ying- Executive Director at IBN.
A big part of the problem is the use of animals or animal-derived cells in preclinical cardiotoxicity studies due to the limited availability of human heart muscle cells. Substantial genetic and cardiac differences exist between animals and humans. There have been a large number of cases whereby the tests failed to detect cardiovascular toxicity when moving from animal studies to human clinical trials*.
Existing screening methods based on 2D cardiac structure cannot accurately predict drug toxicity- while the currently available 3D structures for screening are difficult to fabricate in the quantities needed for commercial application.
To solve this problem- the IBN research team fabricated their 3D heart tissue from cellular self-assembly of heart muscle cells grown from human induced pluripotent stem cells. They also developed a fluorescence labelling technology to monitor changes in beating rate using a real-time video recording system. The new heart tissue exhibited more cardiac-specific genes- stronger contraction and higher beating rate compared to cells in a 2D structure.
“Using the 3D heart tissue- we were able to correctly predict cardiotoxic effects based on changes in the beating rate- even when these were not detected by conventional tests. The method is simple and suitable for large-scale assessment of drug side effects. It could also be used to design personalized therapy using a patient’s own cells-” said lead researcher Dr Andrew Wan- who is Team Leader and Principal Research Scientist at IBN.
The researchers have filed a patent on their human heart tissue model- and hope to work with clinicians and pharmaceutical companies to bring this technology to market.
This finding was reported recently in the Biofabrication journal.

The researchers who created the 3D heart tissue from stem cells for drug testing (clockwise from bottom right: Dr Andrew Wan- Dr Hongfang Lu- Dr Ashley Lim- Dr Jia Kai Lim- Ms Ying Ping Chua and Dr Chan Du).
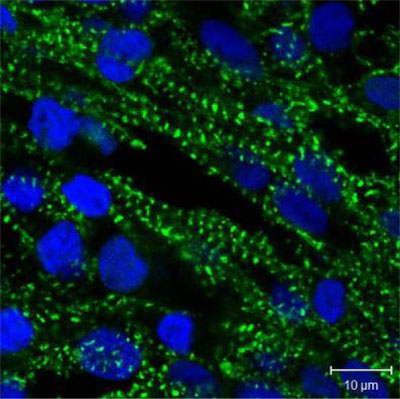
This image shows human heart muscle cells growing in the 3D tissue structure. The cells have been stained with fluorescent molecules to identify the nuclei in blue- and cardiac-specific protein- in green.
Reference:
- H. F. Lu- M. F. Leong- T. C. Lim- Y. P. Chua- J. K. Lim- C. Du and A. C. A. Wan- “Engineering a Functional Three-Dimensional Human Cardiac Tissue Model for Drug Toxicity Screening-” Biofabrication- 9 (2017) 025011. See article.
About the Institute of Bioengineering and Nanotechnology (IBN)
The Institute of Bioengineering and Nanotechnology (IBN) is the world’s first bioengineering and nanotechnology research institute. Established in 2003- IBN’s mission is to conduct multidisciplinary research across science- engineering- and medicine for breakthroughs to improve healthcare and quality of life. IBN’s research activities are focused on Nanomedicine- Synthetic Biosystems- Biodevices and Diagnostics- and Green Chemistry and Energy. The Institute has published over 1-200 papers in leading scientific journals- filed 685 active patents and patent applications on its inventions- and established 12 spin-off companies. To nurture young research talents- IBN runs a Youth Research Program that offers students research attachment opportunities and exposure to biomedical research.For more information on IBN- please visit www.a-star.edu.sg/ibn.
About the Agency for Science- Technology and Research (A*STAR)
The Agency for Science- Technology and Research (A*STAR) is Singapore's lead public sector agency that spearheads economic oriented research to advance scientific discovery and develop innovative technology. Through open innovation- we collaborate with our partners in both the public and private sectors to benefit society.
As a Science and Technology Organization- A*STAR bridges the gap between academia and industry. Our research creates economic growth and jobs for Singapore- and enhances lives by contributing to societal benefits such as improving outcomes in healthcare- urban living- and sustainability.
We play a key role in nurturing and developing a diversity of talent and leaders in our Agency and Research Institutes- the wider research community and industry. A*STAR oversees 18 biomedical sciences and physical sciences and engineering research entities primarily located in Biopolis and Fusionopolis.
* >20% of drug attrition has been attributed to cardiovascular toxicity (Laverty et al.- 2011)
Was This Article Helpful?
A*STAR celebrates International Women's Day

From groundbreaking discoveries to cutting-edge research, our researchers are empowering the next generation of female science, technology, engineering and mathematics (STEM) leaders.
Get inspired by our #WomeninSTEM
