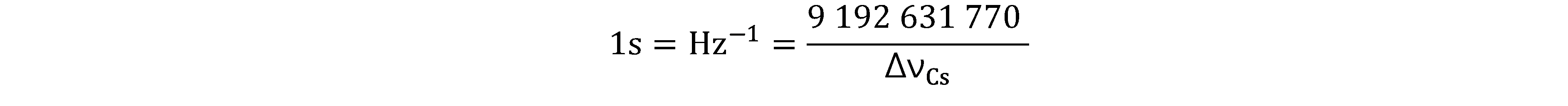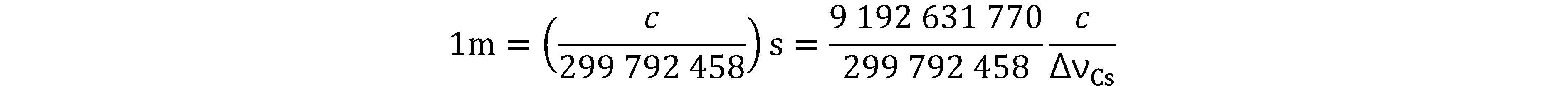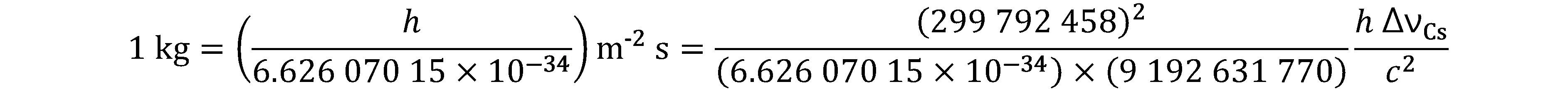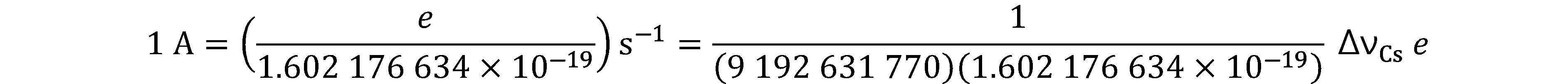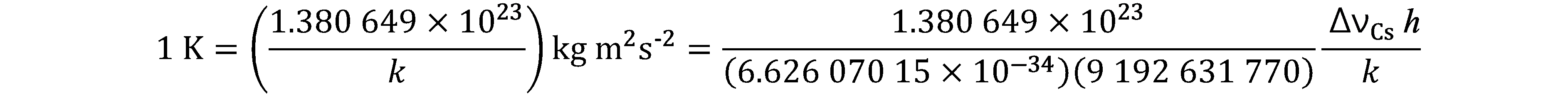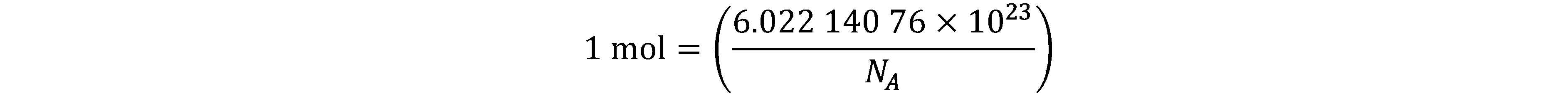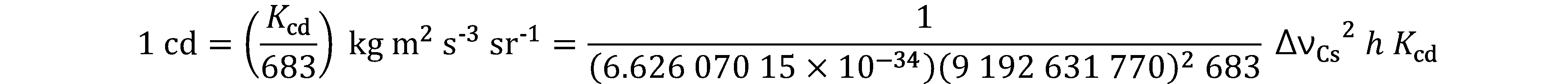SI units Realisation
The International System of Units, the SI, has been widely accepted around the world as the preferred system of units for science, technology, industry and trade since it was established in 1960 at the 11th meeting of the Conférence Générale des Poids et Mesures, the CGPM (the General Conference on Weights and Measures, in English).
On 20 May 2019, came into force a new approach to articulating the definitions of the SI units by fixing the numerical values of seven “defining” constants.
As a signatory to the Metre Convention, Singapore adopts the redefined SI to ensure that we are consistent with international best practice. The National Metrology Cerntre, A*STAR, is the primary agency responsible for implementing the SI definitions in Singapore.
Since the product of the numerical value and the unit has to equal the value of a constant, by fixing the exact numerical value the unit becomes defined. The seven constants are chosen in such a way that any unit of the SI can be written either through a defining constant itself or through products or quotients of defining constants. All SI units are defined in terms of constants that describe the natural world. This will assure the future stability of the SI and open the opportunity for the use of new technologies, including quantum technologies, to implement the definitions.
Prior to the new revision of the SI, SI units have been presented in terms of a set of, most recently seven, base units, namely
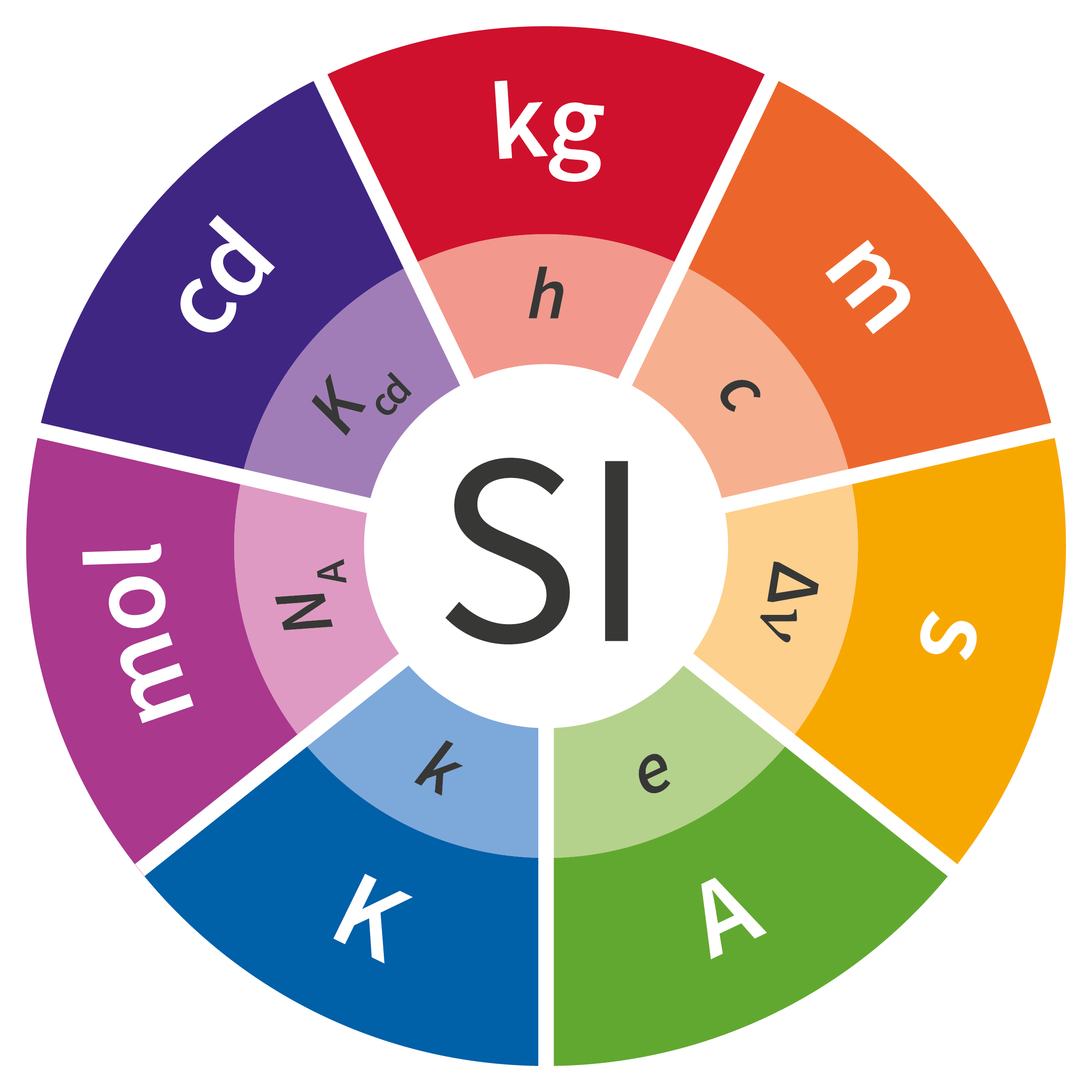
All other units, described as derived units, are constructed as products of powers of the base units. This concept of base and derived units is maintained. With the new revision, the definitions of all the base units can now be deduced by using one or more of these defining constants.
The derived units were constructed as products of powers of the base units. In fact, defining the SI by fixing the numerical values of seven defining constants has the effect that this distinction is, in principle, not needed any more, since all units, base or derived units, can now be written either through a defining constant itself or through products or quotients of defining constants.
The new approach will assure the future stability of the SI and open the opportunity for the use of new technologies, including quantum technologies, to implement the definitions.
Please visit this link for more information on the SI.
A*STAR celebrates International Women's Day

From groundbreaking discoveries to cutting-edge research, our researchers are empowering the next generation of female science, technology, engineering and mathematics (STEM) leaders.
Get inspired by our #WomeninSTEM