Rong En TAY
Biography
Dr Tay Rong En graduated from Harvard University in 2019 with a PhD in Immunology for his work in Professor Kai Wucherpfennig’s group at the Dana-Farber Cancer Institute, where he used functional small molecule and CRISPR screening approaches to identify HDAC3 and CARM1 as novel epigenetic regulators of CD8 T cell effector function. Upon returning to Singapore Immunology Network, he applied his expertise using screening strategies to search for novel immunotherapy targets for hepatocellular carcinoma as a postdoctoral fellow in the groups of Prof Laurent Renia and Dr Olaf Rötzschke, in close collaboration with Prof Toh Han Chong from the National Cancer Centre.
For his work on discovering and validating novel targets for HCC immunotherapy, Dr Tay was awarded a Young Individual Researcher Grant from the National Medical Research Council and a pre-pilot award from the Singapore Therapeutics Development Review. In October 2023, Dr Tay started his own research group as a Group Leader, where he continues his work on hits from his screening approaches in HCC and beyond.
Main Appointments
- Group Leader, Singapore Immunology Network (SIgN, A*STAR, Singapore)
- Visiting Scientist, National Cancer Centre Singapore (NCCS, Singapore)
Research Focus
Our research focuses on the systematic discovery and validation of novel therapeutic targets for immunotherapy of solid tumours using focused functional genetic and pharmacological screens, beginning with hepatocellular carcinoma. Hepatocellular carcinoma (HCC) is the primary form of liver cancer and a highly lethal disease (>900,000 new cases and >800,000 deaths globally every year), with most cases occurring in East and Southeast Asia. HCC is most commonly detected at an advanced stage that is typically fatal within a year if left untreated. Although recent successes with combination immunotherapies have revolutionised treatment options for HCC, about two-thirds of patients with advanced HCC still do not respond to any kind of therapy.
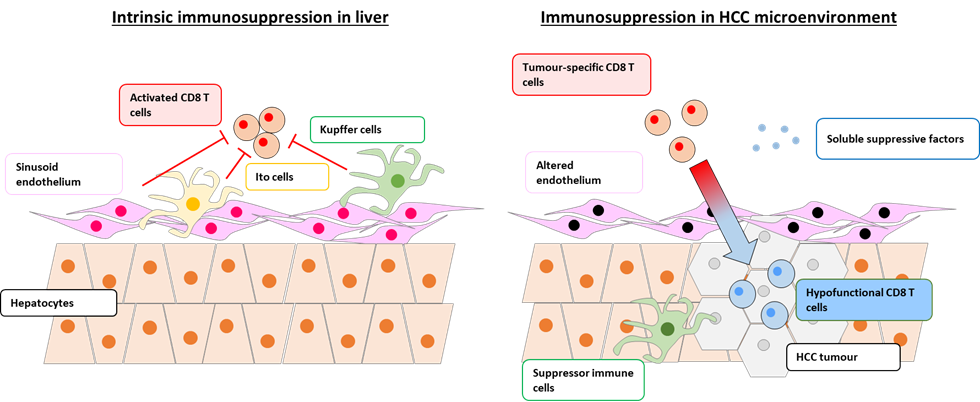
A critical gap in the rational design of the next generation of effective HCC immunotherapeutics is the paucity of knowledge of dominant immunosuppressive molecular pathways specific to the liver and to the HCC tumour microenvironment.
My group uses two approaches to uncover targets:
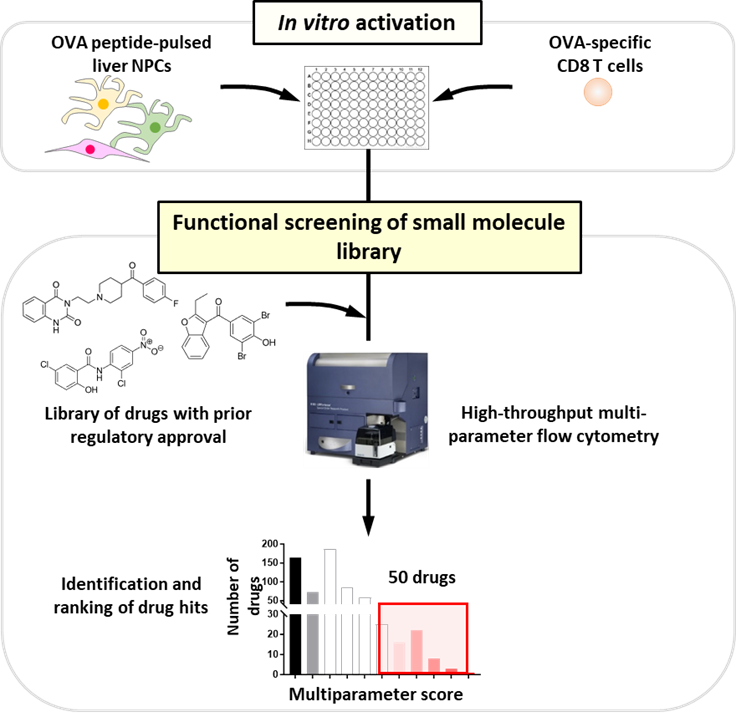
- High-throughput functional phenotypic in vitro screening of drugs for their ability to block the immunosuppression of activated CD8 T cells by liver-resident cells. Click here for an example from my previous work where we used a similar approach to discover a novel role for the epigenetic regulator HDAC3 in regulating CD8 T cell cytotoxicity.
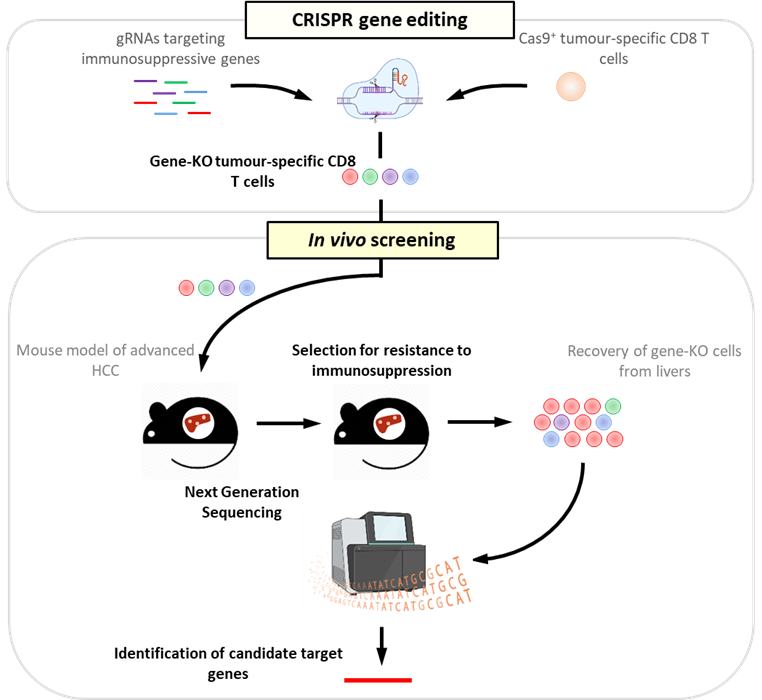
- Focused CRISPR-Cas9 genetic screening of potential target molecules on tumour-infiltrating CD8 T cells in mouse models of HCC. Click here for an example from my previous work where we used a similar approach to identify the epigenetic regulator CARM1 as a potential target for immunotherapy in a mouse model of melanoma that is highly resistant to immunotherapy.
By combining these two approaches, we aim to discover immunosuppressive molecular drivers and pathways intrinsic to the liver and specific to the HCC tumour microenvironment, respectively.
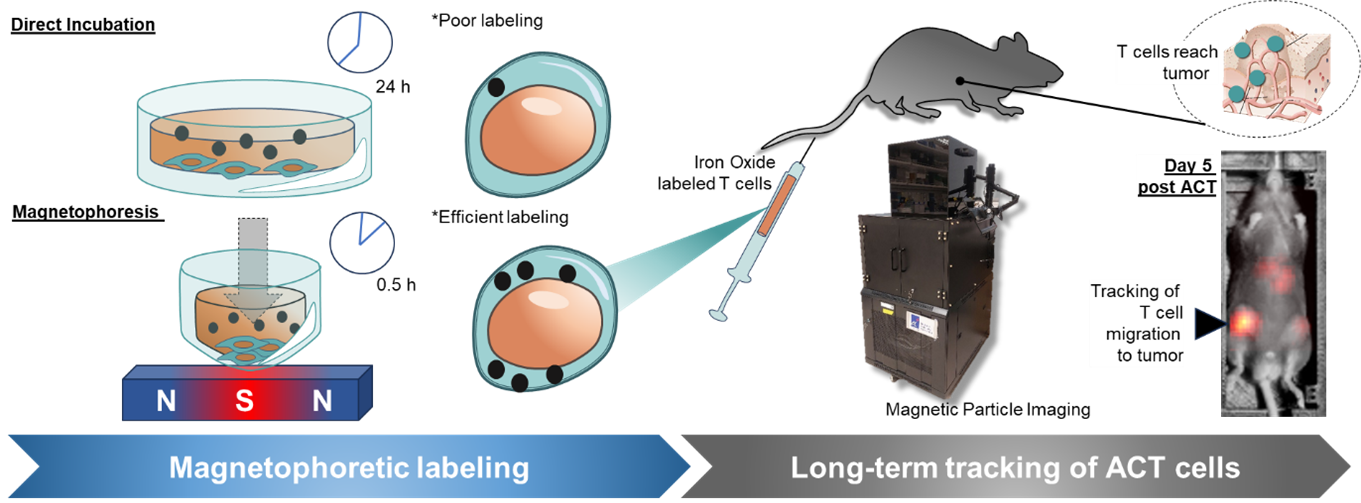
More recently, we have developed a high-throughput platform for magnetic particle imaging (MPI) of adoptively transferred immune cells labelled with magnetic nanoparticles. This platform uses a custom-built MPI device developed by our partner, Dr Tay Zhi Wei (formerly from IBB, currently moved to National Institute of Advanced Industrial Science and Technology, Japan) and is capable of imaging 5 mice in one sitting for at least two weeks following cell transfer, enabling the longitudinal monitoring of their dynamics in real-time and accelerating research in cell therapies for solid tumours.
Overall, our research aims to generate validated, high-confidence targets for the downstream development of rationally engineered therapeutics and cell therapy products in partnership with clinicians driving clinical trials and with industry partners.
Lab Members
(Left to Right) Charmaine, Hui Chien, Rong En, Daniel.
| Postdocs (Ph.D) | Research Officers |
| Daniel LOPEZ | |
| Hui Chien TAY | |
| Chee Mhin Chin |
Publications
Publications_RONG EN TAY (last updated 30 June 2025)
A*STAR celebrates International Women's Day

From groundbreaking discoveries to cutting-edge research, our researchers are empowering the next generation of female science, technology, engineering and mathematics (STEM) leaders.
Get inspired by our #WomeninSTEM