The Biodesign Process
The Biodesign Innovation Process is a robust multi-disciplinary and systematic approach for the identification of important unmet healthcare needs, the development of novel technologies to address them, and the subsequent development of business and commercialization plans to bring them into patient care. In particular, with evolving industry trends, Biodesign is well suited to explore new innovation solutions at the convergence of Healthcare IT, drugs and MedTech.
Biodesign is adapted from the design thinking process which first defines the problem and then implements the solutions, always with the needs of the user demographic at the core of concept development. This process focuses on needs finding, understanding, creating, thinking, and doing. At the core of this process is a bias towards action and creation: by creating and testing something, you can continue to learn and improve upon your initial ideas.
Design Thinking Process
The design thinking process consists of these 5 steps: Empathize, Define, Ideate, Prototype and Test. For Biodesign, which applies design thinking to healthcare, there are 2 added dimensions of regulatory and reimbursement that need to be considered in the context of implementation into a healthcare system.
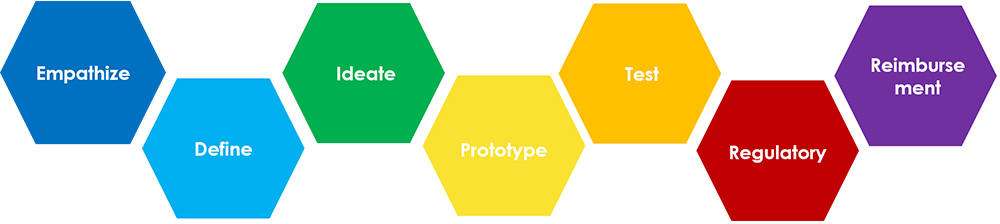
Reference: Design Thinking Process. Adapted from http://web.stanford.edu/group/cilab/cgi-bin/redesigningtheater/the-design-thinking-process/
The process is both iterative and cyclical, going through a flare and focus approach and often requires returning to prior stages and phases as new information becomes available through research.
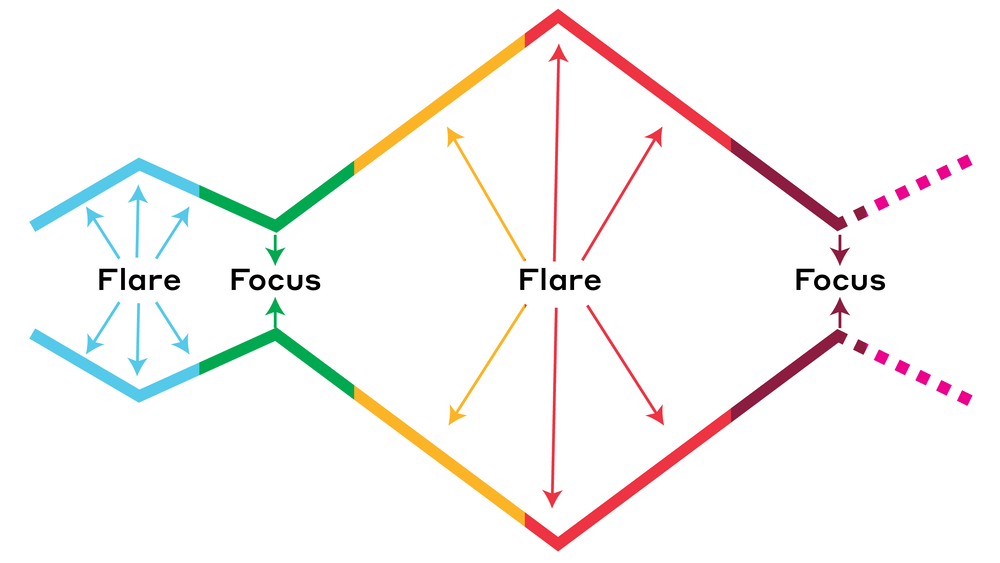
Reference: Necktie Model of Flare and Focus Depicting the Design Thinking Process. Taken from https://dschool.stanford.edu/executive-education/dbootcamp
Biodesign Process
Biodesign is segmented into 3 main phases: Identify, Invent, and Implement with a further deep dive into each sub-segments as follows: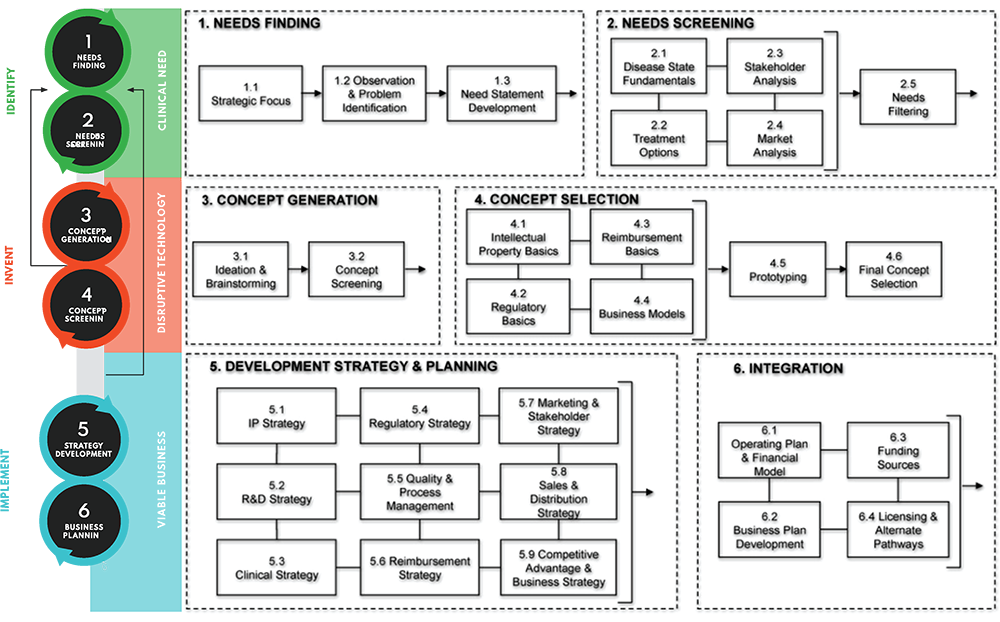
Reference: The Biodesign Process. Adapted from Biodesign: The Process of Innovating Medical Technologies, Yock P. et al., ISBN13: 9781107087354
The Biodesign innovation process starts off with the Identify phase where the team will have to identify an unmet need, preferably through on-the-ground observation in the healthcare setting that can be further validated through interviews with the right stakeholders including patient, payor and provider.
It is important to choose a ‘good’ need with appropriate needs characterization from a multi-disciplinary perspective. With this in mind, we developed our needs-driven mantra as follows:
1. The foundation of an innovation is shaped by starting with a “good” clinical need
2. A well-characterized need is the DNA of a great invention
3. A “good” need looks for insights beyond the immediate technical problem

Reference: Identify Phase: Needs Finding and Screening following the Flare and Focus approach of Design Thinking. Taken from Stanford Byers Center for Biodesign
At the Invent phase, the process will require the team to brainstorm and generate possible concepts without bias. They will follow up with multiple layers of screening to narrow down to the top concept, taking into account the various parallel strategies concerning intellectual property, regulatory, technical feasibility/product development, business model/reimbursement.

Reference: Invent Phase: Concept Generation and Selection following the Flare and Focus approach of Design Thinking. Taken from Stanford Byers Center for Biodesign.
Once the final concept and prototype is created, the 3rd I of the Biodesign process, 'Implement' will commence. At this phase, the team will look into formulating a development strategy and plan to move towards technology commercialization.
Find out more about the Biodesign Process Here.
A*STAR celebrates International Women's Day

From groundbreaking discoveries to cutting-edge research, our researchers are empowering the next generation of female science, technology, engineering and mathematics (STEM) leaders.
Get inspired by our #WomeninSTEM
